Absolute humidity is the mass of water vapor divided by the mass of dry air in a certain volume of air at a specific temperature. The warmer the air is, the more water it can absorb. Absolute humidity is the measure of water vapor or moisture in the air, regardless of temperature. It’s expressed as grams of moisture per cubic meter of air (g/m3).
The maximum absolute humidity of warm air at 30 degrees C (86 degrees F) is approximately 30 grams of water vapor, or 30 g/m3. The maximum absolute humidity of cold air at 0 degrees C (32 degrees F), the freezing point, is approximately 5 g of water vapor or 5g/m3. This would be the differential between humidity in the summer months versus the humidity in the winter months. Warmer air holds more water, whereas colder air holds less water.
Example
As an example, consider two containers:
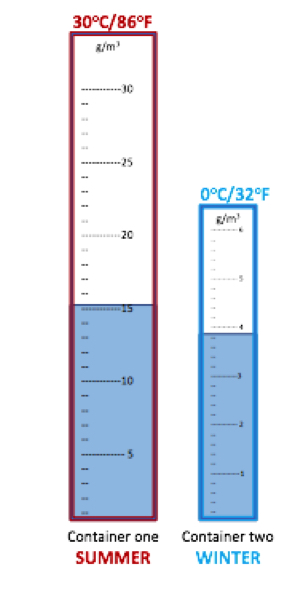 |
| Summer Winter Humidity Comparisons |
Container one has a maximum volume of 30 g of water and is half full, or at 50% of its capacity. Container two has a maximum volume of 5 g of water and is three quarters full, or 75% of its capacity. Container one contains four times as much water as container two, yet actually contains a lower percentage of relative humidity.
If we now call container one ‘summer’ and container two ‘winter,’ we can differentiate between absolute and relative humidity.
Relative Humidity
Relative humidity is the ratio of the absolute humidity at a given time to the highest possible humidity, which depends on current air temperature. Relative humidity doesn’t tell us how much water vapor is in the air, but what percentage of the maximum vapor pressure has been reached.
For example, say the maximum vapor pressure allows a certain volume of air to hold 100 kilograms of water. Supposed this volume of air is only holding 40 kilograms of water. The relative humidity would be 40 kilograms out of 100 kilograms, or 40%, because only 40% of the maximum vapor pressure is being used.


Comments are closed