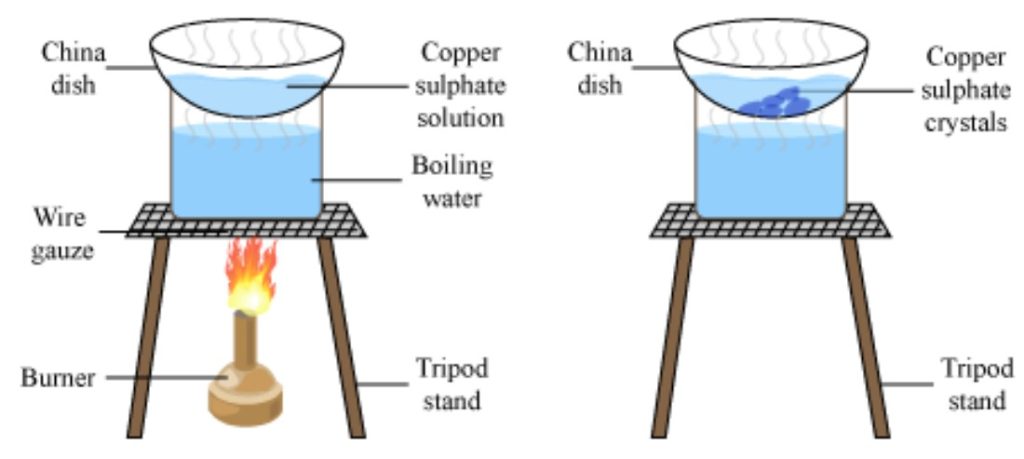
Crystallization is defined as a process by which a chemical is converted from a liquid solution into a solid crystalline state. The widespread use of crystallization within industry is in part due to the fact that crystallization acts as both a separation and purification step; almost all chemical processes utilize at least one crystallization step (either as key separation mechanism or final product engineering).
How does the crystallization process occur?
The crystallization process consists of two major events:
- Nucleation – Molecules gather together in clusters in a defined manner. Clusters need to be stable under current experimental conditions to reach the “critical cluster size” or they will redissolve. It is this point in the crystallization process that defines the crystal structure.
- Crystal Growth – Nuclei that have successfully achieved the “critical cluster size” begin to increase in size. Crystal growth is a dynamic process, with atoms precipitating from solution and becoming redissolved. Supersaturation and supercooling are two of the most common driving forces behind crystal formation.
Development of crystallization processes represents a complex and challenging issue, requiring simultaneous control of various product properties, including purity, crystal size and shape, and molecular level solid structure. The control of the nucleation phase is difficult but is the key to process control; crystallization chemists usually aim to achieve goals of high purity and high yield by solely using controlled cooling crystallization techniques.
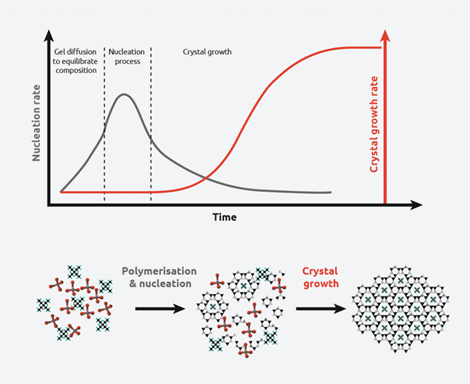
Figure 1: The crystallization process – crystal growth rate vs. nucleation rate
Many compounds can exist in multiple crystal structures – a phenomenon known as “polymorphism” – and can have different physical properties (melting point, shape, dissolution rate, etc.). Depending on the conditions used, either nucleation or crystal growth may be predominant over the other, leading to crystals with different shapes and sizes. Therefore, controlling polymorphism is of significant interest in chemical manufacture.
A common example of the importance of crystal size can be found with ice-cream. Small ice crystals, formed through rapid cooling, improve the texture and taste of the ice-cream compared with larger ice crystals.
Solubility Curves, Supersaturation and the Metastable Zone Width (MSZW)
Traditionally, crystal formation has been achieved by reducing the solubility of the solute in a saturated solution in a variety of ways.
Solubility curves are a common tool used by scientists to understand/demonstrate the relationship between solubility, temperature, and solvent type. By plotting this information, scientists can find the optimum solvent/anti-solvent, temperature, and theoretical yield for their crystallization process.
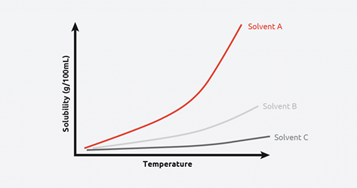
Figure 2: A solubility and temperature graph for various solvents
Figure 2 shows that the given material is highly soluble in Solvent A, meaning more material can be crystallized from a given volume of solvent. Conversely, the given material has a low solubility in Solvent C across all temperatures, potentially making it a good anti-solvent for this material
In reality, when a saturated solution is cooled, there is more solute in the solution than predicted by the solubility curve, and this is referred to as “supersaturation”.
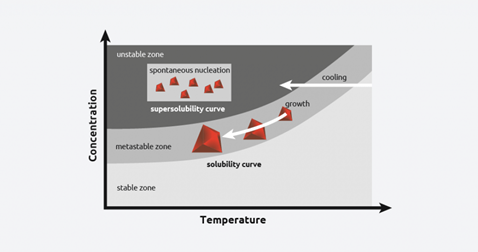
Crystallization solubility curve
As cooling continues, at a certain temperature, crystal nucleation will begin. This temperature is called the “metastable limit”, and the difference between this temperature and the solubility curve is known as the metastable zone width (MSZW).
By carefully controlling the level of supersaturation of a solution, scientists can control the crystallization process.
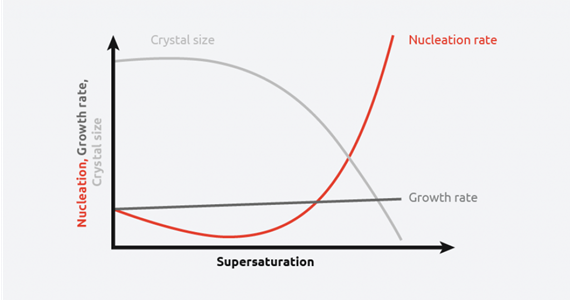
Nucleation, growth rate, and crystal size
As can be seen from the above schematic, at low levels of supersaturation, crystals grow more quickly than they nucleate resulting in large crystal size distribution. At high supersaturation levels, nucleation dominates crystal growth, providing smaller crystals. This makes understanding and controlling supersaturation vitally important when creating crystals of a desired size and distribution.
What role does crystallization play in industrial research, development, and manufacturing?
Crystallization is one of the most widely used technologies in chemical industry, and process robustness governs process productivity and economics. In particular, the pharmaceutical and food sectors are utilizing crystallization for optimized separation, purification, and solid form selection. For example, crystallization is the most common method of formation of pharmaceutical solids for Active Pharmaceutical Ingredient (API) development. The optimization of the particulate properties such as particle size and shape distributions is paramount as the physical form dictates drug product quality and effectiveness.
Many pharmaceutical drugs have poor physiochemical profiles, such as poor solubility in biological fluids. Significant research and development efforts have been made towards developing a solid form landscape that covers all possible solid structures, including polymorphs, solvates, co-crystals, salts, and the amorphous phase to improve Active Pharmaceutical Ingredient (API) development.