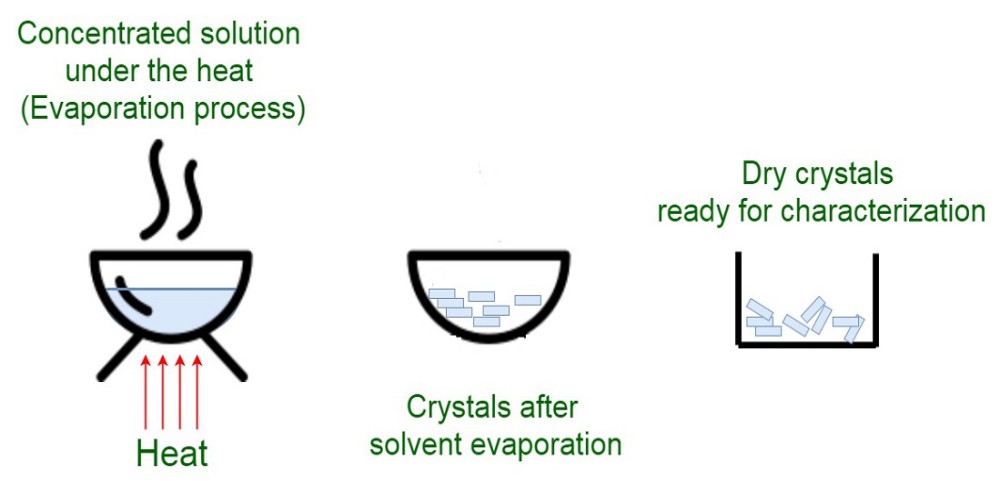
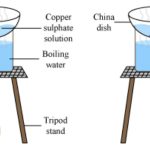
Crystallization is the oldest “unit operation” in a chemical engineering sense. For example, Sodium Chloride has been manufactured this way since the dawn of civilization.
Various traditional methods for crystallization exist, with each technique having unique benefits and drawbacks. The method chosen must be selected based on the properties of the material being crystallized.
- Solvent Evaporation – Easy to set-up, requires air stable samples, requires a minimum solvent volume to work effectively. Large amount of material required.
- Slow Cooling – Requires solvents with boiling points less than 100 °C and moderate solute solubility. Large amount of material required.
- Solvent/Vapour Diffusion – Works well with small amounts of material, however finding two suitable solvents can be challenging. Can “oil out”.
- Sublimation – Not the method of choice for diffraction quality crystals. Typically performed at high temperatures, causing crystals to grow too quickly.
Sonocrystallization
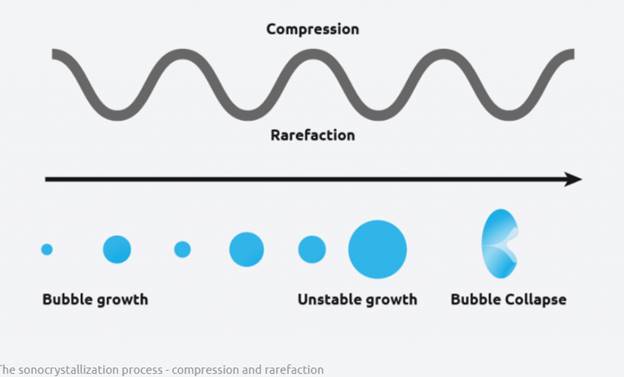
Crystallization processes are often difficult to control, but sonocrystallization is a more modern method of crystallization that offers significant advantages over traditional methods. Ultrasound radiation is known to induce acoustic cavitation in liquids through the formation, growth, and collapse of bubbles. The collapse of the bubble provides energy to encourage the nucleation process at the earliest possible point in time. This results in highly repeatable and predictable crystallizations, and offer various benefits, including;
- Reduced induction time
- A decrease in metastable zone width (MSZW)
- Increased nucleation rate
- Increased crystal growth rate
- Reduce agglomeration
- Tailored crystal size distribution
Discover the Sonocrystallization application page for more information
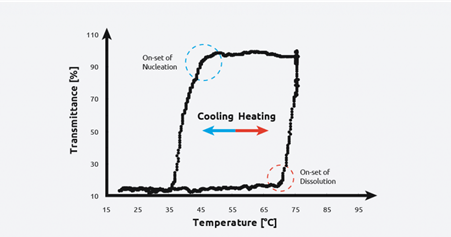
Onset of nucleation and dissolution graph in the sonocrystallization process