‘Little and large’
Perhaps unsurprisingly, the reactor configuration is known to affect the selectivity of oxidation reactions, since it can affect mass transfer due to differences in gas–liquid–solid contacting.47 In terms of efficient heat management, the enhanced heat transfer characteristics of microreactors is particularly valuable, due to the large surface-to-volume ratio of between 10 000 and 50 000 m2 m−3.48 Furthermore, the residence time can be precisely controlled by changing the flow speed or the length of the channels. This has enabled their application to several reactions with the potential for thermal run-away. Successful implementations of hazardous reactions in microreactors are highlighted in recent reviews, including continuous flow oxidation reactions in the liquid phase.11 However, the productivity of microreactors is generally not compatible with industrial production rates. On the other hand, the type of reactors employed in the commodity industry for catalytic oxidation reactions are often dedicated to tonne-scale processes, designed to be operated continuously over the lifetime of the chemical plant. Thus, for speciality chemicals, there is a need for new reactor designs that can overcome the heat management issue, while maintaining high selectivity, ideally offering flexible productivity rates.
In this section, different reactor designs suitable for performing aerobic oxidation reactions at the appropriate scale for fine chemicals and pharmaceutical companies are described, along with their advantages and disadvantages. The different reactors are summarised in Table 2, differing in the way the catalyst (if used) is incorporated in the reactor, as well as the mode of O2 delivery. Homogeneous catalysts can be more selective compared to heterogeneous catalysts, and no internal diffusion resistances exist since they are molecularly dispersed within the reaction medium. However, it can be difficult and expensive to recover the catalyst, since a downstream separation section is required.
Table 2 Types of flow reactors employed for aerobic oxidation reactions
| Homogeneous/no catalysts | Heterogeneous catalysts |
| Segmented flow | Packed bed |
| Membrane | Wall-coated |
| Membrane |
Segmented flow reactors
The segmented flow reactor is easy to construct and it is also the most common system employed for reactions performed with or without a catalyst. In this system, the gas (pure O2 or diluted in N2) is premixed with the liquid in a T-junction followed by a tubular reactor which results in segmented (i.e. Taylor or slug) flow, which is characterised by liquid recirculation inside the slugs (Fig. 3). This ensures good mass transfer of the gas in the liquid. In fact, Taylor flow liquid side volumetric mass transfer coefficients (kLa) up to 10 s−1 have been obtained (for channels smaller than 1 mm), which is typically one order of magnitude higher than traditional contactors such as bubble columns, packed columns etc. Furthermore, such reactors offer efficient heat exchange with the reactor walls, a residence time distribution characteristic of plug flow, and allow easy operation at higher temperature and pressure compared to conventional round bottom flasks.49,50 In many cases Taylor flow is adopted, and this can be ascertained visually due to the transparency of the plastic tubes usually employed. When stainless steel tubes with larger diameters are used, however, the precise flow pattern is much more difficult to establish. Nevertheless, higher velocities of the liquid and gas can be achieved in larger reactors, which can contribute to even better gas–liquid mixing.
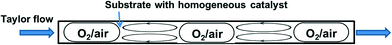
Fig. 3 A segmented-flow reactor (with recirculatory flow patterns).
Supported by a pre-competitive consortium of pharmaceutical companies (Eli Lilly, Pfizer and Merck), a research group led by Stahl at University of Wisconsin-Madison (MadOx Consortium) has been developing effective strategies for aerobic oxidation reactions. In their earlier work, a segmented flow reactor was employed to oxidise various primary and secondary alcohols into aldehydes and ketones using a Pd(OAc)2/pyridine homogenous catalyst.51 A 6.35 mm O.D. (5 mL) stainless steel tube was initially used at the lab scale, while larger tubes were used to produce acetophenone from 1-phenylethanol in near quantitative yield at 25 g and 1 kg scales, respectively. Operation of the 5 mL reactor with a single volume of solution and continuous flow of oxygen (representing batch reaction) provided identical yield as compared with continuous flow of liquid, showing that this reaction could be easily translated to continuous flow. However, batch reaction in a flask equipped with an O2-filled balloon, at lower temperature and pressure, required more than an order of magnitude longer time to reach the same yield. To operate safely, diluted oxygen (8% O2 in N2) was used, which required high total gas pressures (25–35 bar) in order to maintain a sufficiently high O2 concentration. More recently, similar reactors were utilised by the same group for the oxidation of benzylic, aliphatic and activated alcohols to the corresponding aldehydes, catalysed by a (bpy)CuI/TEMPO (2,2,6,6-tetramethyl-1-piperidinyloxyl) catalyst.
Pieber and Kappe used a stainless steel coil (0.8 mm I.D.) for oxidation of 2-benzylpyridines (7) to the corresponding ketones (8) with synthetic air at 200 °C, using inexpensive iron(iii) chloride as a catalyst and propylene carbonate (PC) as a non-toxic and thermally inert solvent (Scheme 5).53 The reaction was enhanced significantly using continuous flow under high temperature and pressure leading to much shorter reaction time than in batch systems. The reactor was replaced with a PFA (perfluoroalkoxy) coil (to prevent corrosion) to perform a variation of the Amoco process, where ethylbenzene was oxidised in the presence of a Co/Mn bimetallic catalyst (Scheme 6). Using 12 bar of synthetic air, the reaction time necessary for complete oxidation of ethylbenzene was 6–7 min, at temperatures 110–120 °C. The acetophenone was formed in 80–84% selectivity, and virtually pure acetophenone was isolated in 66% product yield. The higher pressure and temperature, which were possible in the continuous flow reactor, provided higher selectivity and much smaller reaction time, as compared to batch reaction in a stirred open vial (selectivity to acetophenone ∼74%, at a conversion of ethylbenzene ∼96%, 150 min reaction time, 80 °C). More forcing conditions led to benzoic acid as the major product. It is worth noting that in both of these systems, the need to remove high-boiling PC solvent (Scheme 5), and the low selectivity (Scheme 6), make them unsuitable for industrial applications for the fine and pharmaceutical industries.
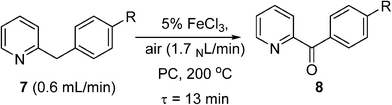
Scheme 5 Aerobic oxidation of 2-benzylpyridines to ketones.
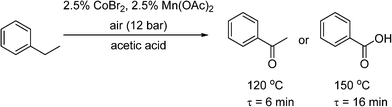
Scheme 6 Oxidation of ethyl benzene to the ketone or benzoic acid.
Two different types of segmented flow reactors have been utilised for manganese-catalysed aerobic oxidation of aliphatic aldehydes to acids. The first was reported by Baumeister et al.,54 who used a 4.7 mL microstructured reactor (one-A Engineering, Austria) for the conversion of valeraldehyde (9a) to valeric acid (10a) with pure oxygen with catalytic manganese(ii) acetate at 40 °C (Scheme 7, eqn (1)). The addition of octanoic acid as a co-feed is essential to maintain higher productivity and selectivity. Generation of gas–liquid interface areas was facilitated by periodic narrowings along the 1 mm I.D. stainless steel reactor channel. Using a large molar excess of oxygen (2 equivalents with respect to the aldehyde), the dominant phase within the reactor is believed to operate in an annular flow pattern. Conversion of 95% at superficial liquid residence time of 82 s with high selectivity (80–85%) was obtained with a high productivity (10 290 kg h−1 m−3). Attempt to scale-up the reactor to include 3 mm channels was unsuccessful.
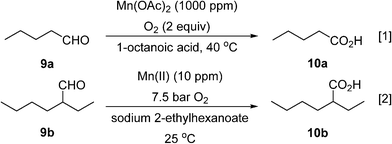
Scheme 7 Oxidation of an aldehyde to a carboxylic acid.
In recent work, Vanoye et al. showed that neat 2-ethylhexanal (9b) can be oxidised safely to the corresponding carboxylic acid (10b) in a PFA tube reactor (1.65 mm I.D.), using pure oxygen at 7.5 bar at ambient temperature and a homogeneous Mn(ii) catalyst (Scheme 7, eqn (2)).55 The system was limited by the low heat transfer coefficient of the PFA which did not allow efficient management of the heat produced by the reaction. This was overcome by lowering the catalyst loading (to 10 ppm) and increasing the O2 pressure to 7.5 bar to maintain 94% selectivity. This was sufficient to maintain a productivity of 130 g h−1 of 2-ethylhexanoic acid, without further purification or solvent separation.
In principle, it is possible to deploy heterogeneous catalysts in segmented flow reactors by suspending catalyst (nano)particles in the reaction mixture, creating a triphasic (gas/liquid/solid) system, as long as the hydrodynamics of the reactor can maintain the catalyst suspension. Further challenges include feeding of liquid/solid slurries and avoiding particle trapping in the reactor or the associated piping. This has been partly demonstrated in a report by Alex et al.,56 who used a PTFE (polytetrafluoroethylene) tube (1 mm I.D.) for the oxidation of benzyl alcohol to benzaldehyde using PVP (polyvinylpyrrolidone)-stabilised Pd and Au/Pd nanoparticles with air. Selectivity of 97% at 91% conversion at a mean residence time of 15 min was obtained at 50 °C and ca. atmospheric pressure under continuous flow (in batch, quantitative conversion with 96% selectivity was achieved under similar conditions at a longer reaction time of 40 min). In this system, the nanoparticles were dispersed in the reaction solution and were precipitated at the end of the reaction. The recovered catalyst could be redispersed for reuse, but showed evidence of deactivation.
Packed bed reactors
In packed bed reactors the heterogeneous catalyst is packed and retained in a particulate form in a cartridge, tube, or microchannel. In this way catalyst separation from the reaction mixture is not required. A packed bed reactor offers easy catalyst replacement in the case of deactivation, or if another reaction needs to be performed. It also allows easier control of reaction (residence) time as compared to a batch reactor, and thus avoiding over-oxidation. In this arrangement, the size of the catalyst particles is typically in the order of 100 μm to avoid excessive pressure drop. The liquid can either be pre-saturated in a plug flow, or fed separately in a trickle bed (Fig. 4). Thus, depending on the conditions, the reactor operates at either triphasic (solid–liquid–gas) or biphasic (solid–liquid) conditions.
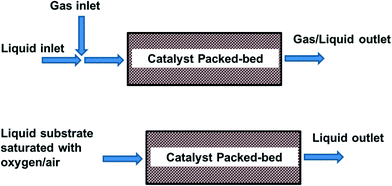
Fig. 4 Packed-bed reactor operating under triphasic (top) or biphasic (bottom) conditions.
For synthetic applications, milliscale packed bed reactors (<10 mm I.D.) are generally favoured. The most common design uses a segmented flow of oxygen/liquid feed through the catalyst bed. This has been used in the assessment of many catalysts (TEMPO,57 Au,47,58 Pd and Pt59) on different supports, typically in the oxidation of benzyl alcohol to benzaldehyde. For example, the MadOx team recently attempted a scale-up of the aerobic oxidation of alcohols using a heterogeneous Ru(OH)x/Al2O3catalyst in a stainless steel tube.46 The liquid and gas (8% O2 in N2) were mixed with a T-piece and fed into the reactor in an upflow direction, operating within the slug flow regime and using tubing with diameter below the flame propagation threshold. Under these trickle-bed conditions, catalyst deactivation was significant. Nevertheless, provided that the operating conditions were adjusted accordingly, high steady state yields in the aerobic oxidation of 2-thiophene methanol could be maintained over 72 h to achieve a large yield of aldehyde product. Reaction rates of various alcohols, as well as deactivation characteristics, were found to be similar in batch conditions. A similar trickle-bed arrangement was also adopted by Kobayashi and co-workers to achieve aerobic oxidation of a number of primary and secondary alcohols with mixed solvents using polymer incarcerated gold-based nanocluster catalysts.60 The catalysts were packed with an optimised amount of Celite in a glass column (0.5 cm I.D.) to prevent obstruction by swelling of the catalysts. In this work, the reactions required the presence of inorganic bases, which were delivered in an aqueous phase in a down flow to the column with the packed catalyst bed, along with a solution of the reactant dissolved in an organic solvent, and O2. Good conversion and selectivity were obtained in a single pass with different catalysts (Au–Pt was found to be selective for aldehydes and ketones in trifluorobenzene, while Au–Pd favoured the formation of methyl esters in methanol), under optimised operating conditions. However, the productivity of the system is too low (space-time-yields up to 9.93 μmol mL−1 h−1) for commercial applications. Comparison with batch systems showed higher selectivities in the flow reactor, because over-oxidation could be prevented by controlling the residence time. To tackle the problem of catalyst deactivation, Muzen et al. described a reactor (4 cm I.D.) operated with ON-OFF liquid flow modulation.61 Gas and liquid streams were fed onto a packed-bed (filled with glass beads) in order to achieve thermal and vapour pressure equilibrium before entering the trickle-bed containing 0.4 kg of Pt/γ-Al2O3. The catalytic oxidation of ethyl and benzyl alcohols can be achieved under mild operating conditions (70 °C). Alcohol conversion can be improved by modulating the split and cycle period.
Mass and heat transfer efficiency of packed-bed reactors can be improved by modifying the catalyst supports. The use of ceramic fibre catalyst supports has received attention in recent years, as they can offer a large-surface-to-volume ratio without compromising on pressure drop. A Ru(OH)x catalyst deposited on a ‘paper-structured’ alumina/silica composite has been described.62 Ten of these disk-shaped porous catalyst disks were stacked in a stainless steel flow reactor, for the selective aerobic oxidation of aromatic and aliphatic alcohols to their corresponding aldehydes and ketones. Better performance was recorded compared to beaded catalysts, which was attributed to the formation of thinner liquid film layers.
Better productivity can also be achieved by improved reactor design. Bavykin et al.employed a multichannel reactor containing static mixers and heat-transfer channels, thus integrating mixing, heat transfer and reaction functionalities.63 The reactor consisted of 5 parallel packed-bed channels of 2 × 2, 3 × 3 and 5 × 5 (mm × mm) cross section and 10 cm length, each preceded by a static mixer to mix the gas and liquid streams before their entry to the catalyst bed. The reactor allowed staged injection of oxygen which was shown to be beneficial due to the development of a more uniform hydrodynamic regime of two-phase flow along the packed reaction channel. The reactor was shown to operate isothermally despite the significant heat formation from the exothermic reaction. In this work, Ru/Al2O3 was used as catalyst for oxidation of benzyl alcohol. Yields up to 55% and selectivity of 99% were achieved in a single pass conversion.
Another way to improve the safety in catalytic packed bed reactors is to saturate the substrate solution with oxygen before reaching the catalyst packed bed. The solubility of oxygen may pose a limitation; for this reason, this approach is only suitable for dilute solutions and high pressures. This was first described by Zotova et al. using a commercially-available X-Cube™ reactor,32 consisting of a stainless steel cartridge packed with Ru/γ-Al2O3. The mobile phase was saturated with O2 at different pressures (5–25 bar) before passing it through the catalyst bed. Using the device, a variety of primary and secondary alcohols could be converted to their corresponding carbonyl compounds in good yields and high selectivities. The system could be operated safely under continuous recirculation, and the turnover frequency was comparable to that achieved with other flow reactors. More recently, such reactor was also utilised by Osako et al. for aerobic oxidation of alcohols in water catalysed by Pt nanoparticles dispersed in an amphiphilic polystyrene–poly(ethylene glycol) resin;64primary and secondary alcohols including aliphatic, aromatic and heteroaromatic alcohols were efficiently oxidised at 40–70 bar. Similarly, an H-Cube™ reactor fitted with an external gas module was used to oxidise benzyl alcohol using Fe/Al-SBA15, with TEMPO as a co-catalyst.65 Conversions of up to 42% in a single pass could be achieved with high selectivity, while continuous recirculation was used to obtain full conversion.
In packed bed reactors, temperature control is usually implemented by external heating elements. An interesting packed bed reactor was described by Kirschning and co-workers,66 where heating was applied inductively directly to the catalyst particles (Au supported on Fe3O4-containing core with SiO2 shell particles) contained within a polyether ether ketone (PEEK) tubing. The liquid phase was pre-saturated with gas by the use of a tube-in-tube AF-2400 device (see section 4.5 below) before entering the packed-bed. The system was used to oxidise allylic and benzylic alcohols where, in almost all cases, full conversion was achieved in a single pass. The system was scaled up to convert 2.5 g of 4-bromobenzyl alcohol to its corresponding aldehyde, with no over-oxidation to the acid. However, the productivity was quite low (operating at a flow rate of 0.2 mL min−1), and catalyst deactivation due to leaching was also observed at the beginning of the process. The same catalyst gave very low conversion of 4-bromobenzylalcohol, when tested under batch conditions in a sealed vial using an external oil bath, possibly due to inefficient oxygen mass transfer.
Catalytic wall reactors
The greatest advantage of using packed bed reactors is that (deactivated) catalysts can be easily replaced. However, there are also certain disadvantages; notably, heat and mass-transfer limitations when reactions are particularly exothermic or fast (as is the case for most oxidation reactions). Another limitation is the need to employ catalyst particles of a certain size, so as not to cause an excessive pressure drop. Incorporating the catalyst into the reactor wall (Fig. 5) allows better mass/heat transfer characteristics for fast reactions.
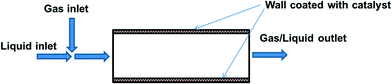
Fig. 5 Catalytic wall reactor.
This was demonstrated in a microreactor, where a gold catalyst was immobilised onto a polysiloxane-coated capillary through cross-linking with a copolymer.67 O2, substrate, solvent (dichloroethane) and aqueous K2CO3 were pre-mixed, and the multiphasic mixture was passed through the capillary tube at 60–70 °C. The Au-based system was used for oxidation of benzylic, aliphatic, allylic, secondary benzylic alcohols to ketones, while a Au/Pd-immobilised capillary column reactor was required for the oxidation of primary benzyl alcohols. The system could be operated continuously for at least four days without loss of activity. The same concept can be applied at a larger scale using a monolith reactor, such as that described by Pollington et al. for the selective oxidation of glycerol.68 Employing a stainless steel reactor (2.5 cm I.D.) housing Au/C coated monoliths, it was designed for co-current downflow operation, liquid recirculation and continuous air feed; with gas and liquid mixed before the catalyst bed. Reaction rate was found to be an order of magnitude greater for the monolith as compared to an autoclave batch reactor.
Membrane reactors
So far in our discussion, all the reactor systems deploy mixtures of O2 and substrate as mobile phases. This may pose safety concerns, particularly if the outlet of the reactor contains gas/vapour mixtures that are within the flammable regime (see section 3.4). Another approach to improve safety is to keep the reactive O2 separate from the other reaction components by using a membrane (Fig. 6), i.e. the membrane acts as a gas distributor.45 Oxygen diffuses across the membrane into the liquid phase, which may already contain the catalyst (homogeneous), or flows into a packed bed (heterogeneous catalyst). Provided that the membrane is chemically inert to organic solvents, this approach allows oxygen to permeate along the length of the reactor, hence avoiding axial concentration gradients. In order to deliver O2 sufficiently quickly without forming a headspace in the reactor, the pressure difference between the two sides needs to be carefully controlled. This will naturally place an upper working limit on the O2 pressure and its subsequent mass transfer into the liquid phase.
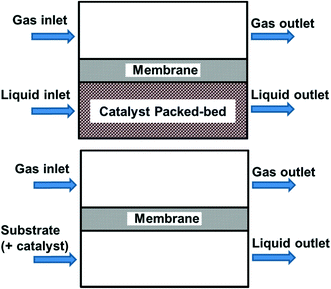
Fig. 6 Membrane reactors separating reactive gas from the other reaction components, using a heterogeneous catalyst (top) or homogeneous/no catalyst (bottom).
One such design used a ceramic membrane with a concentric configuration to minimize the radial packed bed mass transfer distance, as demonstrated by Constantinou et al.70 In this work, an inner tube created an annulus for the catalyst packed-bed through which the liquid phase flowed. The tubular membrane (7 mm I.D.), comprised of layers of alumina and a zirconia top layer with an average pore size of 50 nm and separated this solid–liquid mixture from O2, which was fed from the opposite side of the membrane in the outer shell of the reactor. Conversion of benzyl alcohol to benzaldehyde was achieved using a Pd–Au/TiO2 catalyst. In order to overcome the deficiency of oxygen in the catalyst bed area (which could lead to lower selectivity), O2 supply was increased by raising the gas pressure, diluting the substrate concentration and increasing residence time, achieving benzaldehyde selectivity 88% at 75% conversion.
If the reaction temperature is sufficiently low, polymeric membranes can be used. A tube-in-tube reactor based on a gas-permeable Teflon AF-2400 membrane was first developed by the Ley group at Cambridge University, initially for homogeneous and heterogeneous catalytic hydrogenation reactions.71 This was subsequently deployed for an anti-Markovnikov Wacker oxidation of functionalized styrenes (11) to linear arylacetaldehydes (12) using PdCl2/CuCl2 catalysts (Scheme 8). Exploratory experiments were performed in a batch reactor, but optimisation of the reaction conditions was carried out in a tube-in-tube reactor (the annulus between the inner and the outer tubes was pressurized with O2) followed by a heated stainless steel reactor coil.72 Although the oxygen permeability of Teflon AF-2400 is high,73 long residence times were required to achieve good yields (56–80%). As O2 has an upper solubility limit in the reaction solvent, O2 depletion is a significant issue at higher reactant concentrations. This may be circumvented by double dosing the reaction stream (by inserting another tube-in-tube device between two reaction coils). This can lead to some improvement, but increasing the O2 pressure can lead to over-oxidation, hence careful adjustment of concentrations is required. In another study by Wu et al.,74an AF-2400 membrane was used to deliver O2 along the entire length of the reactor packed with Pd-Au/TiO2 catalyst particles, for the aerobic oxidation of benzyl alcohol (known to be a very fast process). At 120 °C and 6 bar of O2, 44% conversion of the neat alcohol could be achieved at 115 gcat s galcohol−1 catalyst contact time. Under these conditions, formation of toluene was a competitive process.
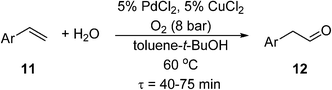
Scheme 8 Anti-Markovnikov Wacker oxidation.
In a search for more cost-effective membranes, cheaper polymeric materials were evaluated by the MadOx group, who found that PTFE exhibited an acceptable combination of low cost, chemical stability and gas diffusion properties. A tube-in-shell reactor was duly constructed with a Teflon coil (1.6 mm I.D.), contained within an oven (tube-in-shell).75 The use of this reactor was demonstrated in the aerobic oxidation reactions of alcohols facilitated by both homogeneous (Cu/TEMPO and Cu/ABNO) or heterogeneous (Ru(OH)x/Al2O3) catalysts. Complete conversion of benzyl alcohol to benzaldehyde (0.5–1 M) can be obtained using the Ru catalyst, but required a residence time of nearly 1 h (9.2 bar and 80 °C). Conversely, homogeneous Cu/TEMPO and Cu/ABNO catalysts were mixed with substrates and flowed within the inner tube, while O2 was pressurised in the outer tube. Near quantitative product yields in a residence time of 1 min were achieved for various benzylic alcohols at 24 bar O2pressure.
Catalytic membrane reactors
A catalytic membrane reactor essentially combines features of a membrane reactor and a catalytic with a catalyst wall reactor. In this design, the catalyst is embedded within a nanoporous membrane, thus positioning it between the liquid–gas boundary, so as to maximise mass transfer whilst also keeping gaseous oxidant and liquid hydrocarbons separated and thus improve safety. Typically, ceramic membranes resembling common catalyst supports (e.g. alumina, silicates) are impregnated with the catalyst. To date, catalytic membrane reactors have been largely employed for gas-phase reactions, and none has yet been reported for aerobic oxidation reactions in the liquid phase. In related work, several membranes were evaluated for direct synthesis of hydrogen peroxide,76 with a Pd catalyst deposited into the finest porous layer on the inner side of the membranes. Oxygen was fed from the outer side of the membrane, while hydrogen was dissolved in methanol solvent at high pressure and fed through the inner side of the membrane. As might be expected, the diffusive transport of the reactants to the catalytically active zone, located on the inner walls of the membrane channel, was found to be crucial. Radial mixing was increased by filling the membrane with small glass beads and led to higher productivity.

Comments are closed