Solvent Criteria
One of the most important steps in developing a successful (economical) extractive distillation sequence is selecting a good solvent. Approaches to the selection of an extractive distillation solvent are discussed by Berg, Ewell et al. ,and Tassions. In general, selection criteria include the following :
1. Should enhance significantly the natural relative volatility of the key component.
2. Should not require an excessive ratio of solvent to nonsolvent (because of cost of handling in the column and auxiliary equipment.
3. Should remain soluble in the feed components and should not lead to the formation of two phase.
4. Should be easily separable from the bottom product.
5. Should be inexpensive and readily available.
6. Should be stable at the temperature of the distillation and solvent separation.
7. Should be nonreactive with the components in the feed mixture.
8. Should have a low latent heat.
9. Should be noncorrosive and nontoxic.
Naturally no single solvent or solvent mixture satisfy all the criteria, and compromises must be reached.
Solvent Screening
Perry’s handbook serve as a good reference for the solvent selection procedure, which can be thought of as a two-step process, i.e.:
Broad screening by functional group or chemical family
1. Homologous series : Select candidate solvent from the high boiling homologous series of both light and heavy key components. Â
2. Robins Chart: Select candidate solvents from groups in the Robbins Chart (part of the chart is shown in Table 3) that tend to give positive (or no) deviations from Raoult’s law for the key component desire in the distillate and negative (or no) deviations for the other key.
3. Hydrogen-bonding characteristic: are likely to cause the formation of hydrogen bonds with the key component to be removed in the bottoms, or disruption of hydrogen bonds with the key to be removed in the distillate. Formation and disruption of hydrogen bonds are often associated with strong negative and positive deviations, respectively from Raoult’s Law.
4. Polarity characteristic: Select candidate solvents from chemical groups that tend to show higher polarity than one key component or lower polarity than the other key.
Identification of individual candidate solvents
1. Boiling point characteristic: Select only candidate solvents that boil at least 30-40oC above the key components to ensure that the solvent is relatively nonvolatile and remains largely in the liquid phase. With this boiling point difference, the solvent should also not form azeotropes with the other components.
2. Selectivity at the infinite dilution: Rank the candidate solvents according to their selectivity at infinite dilution.
3. Experimental measurement of relative volatility: Rank the candidate solvents by the increase in relative volatility caused by the addition of the solvent.
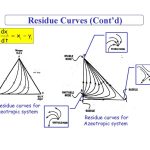
Residue curve maps are of limited usefulness at the preliminary screening stage because there is usually insufficient information available to sketch the them, but they are valuable and should be sketched or calculated as part of the second stage of the solvent selection.
Comments are closed