To understand the fundamental chemistry of silver-based photography, we must look at the photochemistry of silver salts. A typical photographic film contains tiny crystals of very slightly soluble silver halide salts such as silver bromide (AgBr) commonly referred to as “grains.” The grains are suspended in a gelatin matrix and the resulting gelatin dispersion, incorrectly (from a physical chemistry standpoint), but traditionally referred to as an “emulsion,” is melted and applied as a thin coating on a polymer base or, as in older applications, on a glass plate.
Figure 1 shows a schematic representation of the silver halide process. When light or radiation of appropriate wavelength strikes one of the silver halide crystals, a series of reactions begins that produces a small amount of free silver in the grain. Initially, a free bromine atom is produced when the bromide ion absorbs the photon of light:
 Ag+Br– (crystal) + hv (radiation) –> Ag+ + Br+ e–
The silver ion can then combine with the electron to produce a silver atom.
Ag+ + e– –> Ag0
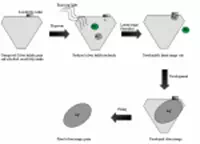 |
| Figure 1: A Simplified SchematicRepresentation of theSilver Halide Process |
Association within the grains produces species such as Ag2+, Ag20, Ag3+, Ag30, Ag4+, and Ag40. The free silver produced in the exposed silver halide grains constitutes what is referred to as the “latent image,” which is later amplified by the development process.
 The grains containing the free silver in the form of Ag4º are readily reduced by chemicals referred to as “developers” forming relatively large amounts of free silver; that deposit of free silver produces a dark area in that section of the film. The developer under the same conditions does not significantly affect the unexposed grains.
The radiation or light sensitivity of a silver halide film (referred to in the trade as its “speed” and denoted on commercial film as its ASA in the United States or DIN in Europe) is related to the size of the grain and to the specific halide composition employed. In general, as the grain size in the emulsion increases, the effective light sensitivity of the film increases – up to a point. An optimum value of grain size for a given sensitivity is found to exist because the same number of silver atoms are needed to initiate reduction of the entire grain by the developer despite the grain size, so that producing larger grains reaches a point of diminishing returns and no further benefit is obtained.
All photographic emulsions contain crystals of varying sizes, but within a given emulsion the range is from less than 0.1 micron in slow emulsions (e.g., for paper prints) to a few microns in “fast” negative emulsions.
An interesting modern innovation in photographic emulsion technology is related to the basic concept of silver halide grain geometry. In a classical silver halide crystal, typically a cubic crystal lattice, the structure will be relatively symmetrical in that the orientation of the crystal in the coated film will always present the same approximate surface area to be exposed. Extensive research efforts led to the development of grain precipitation processes that produced flatter “tablet” grains in which the crystals possessed a more asymmetric geometry, and in which a larger surface area was presented for exposure for the same given weight of silver halide (Fig. 2). That development resulted in significant improvements in film sensitivity and reductions in the amount of silver needed to obtain a given sensitivity – and a potentially important reduction in the cost of the film.


