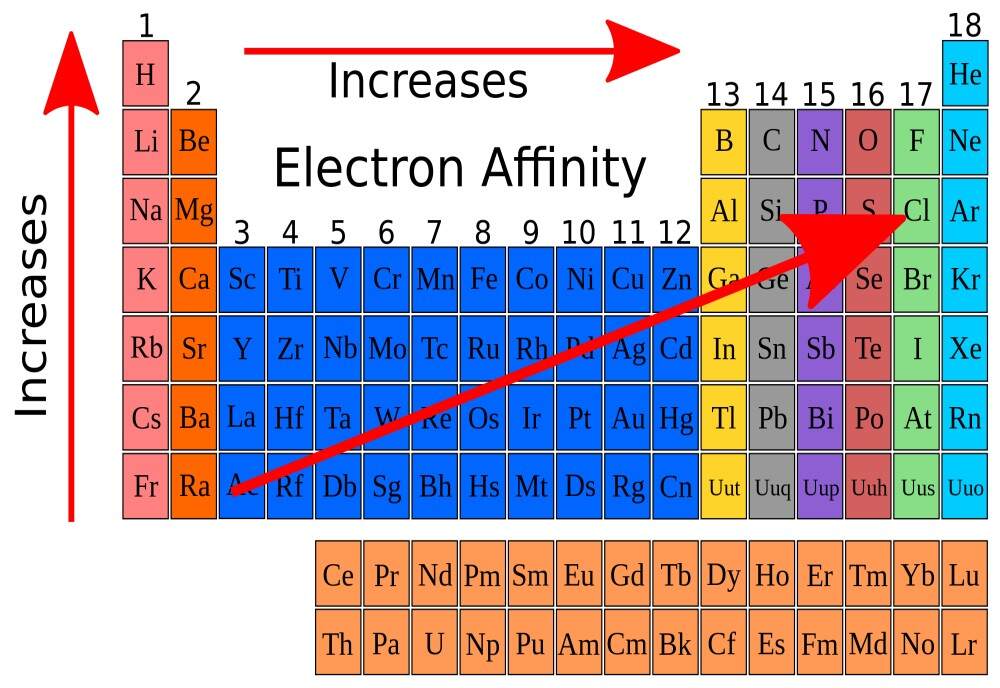
Gases such as helium, neon and argon are so unreactive that we Krypton, xenon and radon will form a very limited number of compounds, e.g. with fluorine, but only under quite exceptional conditions. call them the inert gases. They form no chemical compounds, and their only interactions are of the London dispersion force type. They cannot form hydrogen bonds, since they are not able to bond with hydrogen and are not electronegative. The outer shell of the helium atom is full and complete: the shell can only accept two electrons and, indeed, is occupied by two electrons. Similarly, argon has a complete octet of electrons in its outer shell. Further reaction would increase the number of electrons if argon were to undergo a covalent bond or become an anion, or would decrease the number of electrons below the ‘perfect’ eight if a cation were to form.
There is no impetus for reaction because the monatomic argon is already at its position of lowest energy, and we recall that bonds form in order to decrease the energy. Sodium atoms always seek to lose a single electron to form the Na+ monocation, because the outer valence shell contains only one electron – that is why we assign sodium to Group I(a) of the periodic table. This single electron helps us explain why it is so favourable, energetically, to form the Na+ cation: loss of the electron empties the outer shell, to reveal a complete inner shell, much like removing the partial skin of an onion to expose a perfectly formed inner layer. So, again, removal of sodium’s single outer electron occurs in order to generate a full shell of electrons. But if we look at an element like magnesium, there are several ionization processes possible:

The energy change in reaction (1) is called the first ionization Care: do not confuse the symbols for molecular iodine I2 and the second ionization energy I(2). Hint: note carefully the use of italic type. energy and the energy associated with reaction (2) is the second ionization energy. We symbolize the two processes as I(1) and I(2) respectively. The second ionization energy is always larger than the first, because we are removing a negative electron from a positively charged cation, so we need to overcome the attractive force between them. The value of I(1) for a magnesium atom is 734 kJ mol−1 , but I(2) for removing an electron from the Mg+ monocation is 1451 kJ mol−1 .
Both ionization energies are huge, but I(2) is clearly much the larger. Table 2.8 contains many other ionization energies for elements 1–10. It is clear from Table 2.8 that each ionization energy is larger than the one before. Also note that the last two ionization energies of an element are always larger than the others. The sudden rise follows because the last two energies represent the removal of the two 1 s electrons: removal of electrons from the 2s and 2p orbitals is easier.