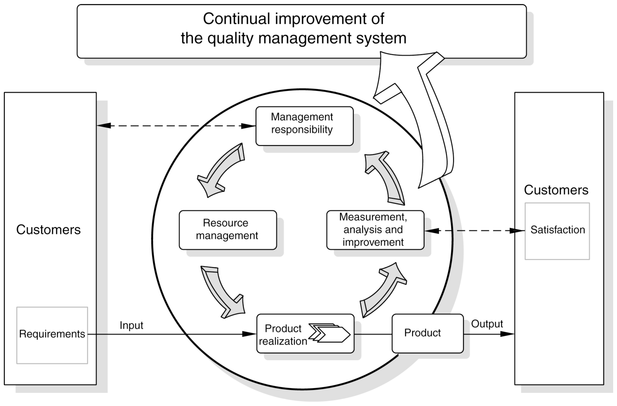
Clause 4.1 CLAUSE 4.1.1 “QUALITY POLICY”
To comply with this requirement there must be a written Quality Policy for the organization. This Quality Policy document must be signed by the current management for the location or site in question. It must also be ensured that the Quality Policy, the objectives and the statement of intent are established and identified in the Quality Manual. The following criteria must be fulfilled: The company has a brief but comprehensive Quality Policy.
This Quality Policy should be included in the Quality Manual as well as being on public display. Quality Objectives have been established by management and there is a statement outlining the intent to deliver against these objectives. The company must ensure that: The Quality Policy is communicated throughout the organization. All employees, at all levels, receive the same message from management. All statements relating to quality that are in documents, training manuals, advertising literature, etc. must be consistent with the Quality Statement.
Any changes must be issued in a controlled manner and must be properly communicated to all employees. It is now necessary for companies to clearly establish goals and appraise the needs and expectations of customers. As the goals will change and evolve these could be documented and expressed in a quality improvement plan which must be thoroughly examined at management reviews. (discussed below).
CLAUSE 4.1.2.1 “ORGANIZATION: RESPONSIBILITY AND AUTHORITY”
To satisfy this requirement the company must ensure that: The Quality Manual contains an Organizational Chart. The Organizational Chart is current, authorized and issued as a controlled document. The management function responsible for quality is clearly identified. The responsibilities and authority of the Quality function are clearly defined. Responsibility and authority under this clause have been extended to cover product processes and quality system non-conformances. This now places greater emphasis on internal quality system activities such as 1 of 10 9/28/2009 1:28 PM internal quality audits, corrective/preventative actions and management review.
CLAUSE 4.1.2.2 “ORGANIZATION: RESOURCES”
The company must ensure that: Adequate resources are provided to allow the effective and efficient operation of the Quality Management system. All personnel involved in the verification of the quality of work have sufficient training and resources to allow them to carry out their duties. All operations involved in the verification of work are identified.
Training records are maintained to provide evidence demonstrating that suitable training has been provided. Training records are treated as Quality Records and managed as such. Establish a clear division between the responsibility for the work and the responsibility for performing assessments. Until recently it was normal to have these different tasks carried by different groups reporting to different managers but there is no reason why self checking cannot be employed with the same person performing both roles but work must also be subjected to independent verification. Adequate training is provided to do both roles and keep the roles separate. Resources required must be adequately identified in the quality system: for example Human Resources will be identified under clause 4.18 (Training) and Calibration under clause (4.11)
CLAUSE 4.1.2.3 “MANAGEMENT REPRESENTATIVE”
The company needs to ensure that: A member of the management team is clearly identified as the person with responsibility for implementing and maintaining the requirements of ISO 9000. This authority is formally defined by some method such as job description, organizational chart or Quality Manual. The management representative must be a member of the management team. A formal mechanism must exist to facilitate the management representative to report to management on system, process or product failures with a view to continually improving the quality system. The standard suggests that the Management Representative be the liaison person with external parties.
CLAUSE 4.1.3 “MANAGEMENT REVIEW”
According to ISO 9002 management are required to assess the suitability and effectiveness of the quality system, at regular intervals, so that they may identify areas where improvements can be made. It is suggested that: All affected groups are represented at management review meetings. The review process is described in the Quality Manual. Minutes of meetings are kept, to show what decisions were made and who was responsible for their implementation. Reviews are held at regular intervals (at least once per year).
Evidence needs to be provided for external auditors to demonstrate that reviews are planned, effective and that corrective action decisions are made and implemented. It should be noted that the last section of this particular ISO 9002 requirement specifies that records of management reviews are required. Management review records should be treated as quality records and managed at such. The review must now, in addition to assessing the continuing suitability and effectiveness of the system, determine whether the stated quality objectives have been achieved. This can only be done by agreeing objective targets for quality system improvements and assessing if these have been achieved.
Clause 4.2 covers the quality system of the organization and it’s documentation. Initially the system organization is dealt with and recognized to be part of TQM (Total Quality Management). Output from each function or part must meet agreed customers’ requirements, be they internal or external. The quality system is understood to be interrelated aspects of: Suppliers’ needs and interests. Customers’ needs and expectations. Therefore a well-planned and managed quality system has to be defined and documented, which are dealt with by quality plan and quality manual sub-clauses.
The quality system and plans should be documented in enough detail so that suppliers and customers can understand it and audit the quality system themselves. This clause requires that the different parts of the company recognize that they are both customers of and suppliers to other parts of the organization as well as to external customers. Sub-clause 4.2.1 now makes a Quality Manual an essential part of the quality system it also requires that other documented procedures are referenced from this document.
This means that a Quality Manual is now mandatory and must reference all the operating procedures either directly from the text or through a cross reference matrix or table. Sub-clause 4.2.2 requires that procedures are consistent with our Quality Policy and should reflect the complexity of the work/processes and skills/training required. The documented procedures must be relevant to the stated quality policy. Where a company can demonstrate through adequate training or other records the acquisition of skills/training by all relevant personnel, detailed procedures are not mandatory; for example personnel who have achieved qualifications through apprenticeship schemes may not require detailed procedures or work instructions. Sub-clause 4.2.3 requires a company to undertake quality planning as appropriate to the organization and that this should be defines and documented.
Clause 4.3 This is the clause that the majority of companies have most difficulty with. Some make the mistake of assuming that this clause applies to the relationship between the company and it’s vendors when in fact it applies to the relationship between the organization’s front end and clients/customers. The 1994 version of ISO 9002 places a much greater emphasis on Contract Review. Where the sales of product and services are covered by contracts there must be clear evidence of a review of terms and conditions. Attention should be paid to the following: Ensuring requirements are agreed and clearly understood by all involved parties. Checking that all parties have the necessary resources, organization and facilities.
VERBAL CONTRACTS
Verbal contracts must be agreed before acceptance implying that some positive confirmation of order requirements must be made. In practice this could be achieved using fax confirmation messages or logging verbal orders and confirming them back to customers over the phone. AMENDMENT TO A CONTRACT This is a new addition to the ISO 9002 requirement for contract review.
The company must have a mechanism for internal communication of all contract amendments … this could be achieved by reviewing ongoing contracts at the daily production meetings by putting a system in place whereby Scheduling, Planning and Production would be alerted to all changes. Clause 4.4 Clause 4.5 This requirement has been updated in the 1994 version (operational since 1 March, 1995.)
DOCUMENT AND DATA CONTROL
Clause 4.5 deals with how documentation should be treated. All documents in must be checked before issue or re-issue after revision. Documents must be positioned so that all persons concerned have easy access to them. A master record file has to be kept. People responsible for revision and issuing of documentation must be identified. Sub-clause 4.5.1 clause requires that documents of external origin such as standards and customer drawings are controlled. Sub-clause 4.5.2 requires that all obsolete documents be adequately identified and that all such documents which are held for reference purposes should be suitably identified. NOTE 10 of ISO 9002 states that documents can be in the form of any type of media such as hard copy or electronic media. If the system is totally electronic then there has to be sufficient availability of hardware (terminals, etc.) to facilitate personnel in accessing procedures, work instruction and records. The procedure for document control must clearly identify who has authority to authorize read and / or write access to controlled documents.
All electronic document and data systems must have adequate security / backup procedures. Clause 4.6 Clause 4.6 deals with purchasing. In the procurement of materials or external services, quality need to be assessed in respect to agreed specifications. Assessment of subcontractors (i.e. vendors or suppliers), ensures that all have the capability of supplying materials or services of the required quality. A formal assessment of capability is needed, possibly through an audit of a subcontractor’s quality or by way of other evidence. Documented evidence on formal assessments is required.
Purchasing data when placing orders should amount to a formal procedure that ensures all necessary information is given to a vendor. Communication links are a central here and a record of performances is needed. Sub-clause 4.6.4.1 “Supplier Verification at Subcontractor’s Premises” This sub-clause requires that all details in relation to product verification and the method of product release by the supplier at the subcontractor’s premises is described on the purchasing documents.
Clause 4.7 Control of Customer-Supplied Product Clause 4.7 deals with purchaser supplies. Here, purchaser means our external customers. In manufacturing the external customer may make “Free Issue”, which is where they provide materials for incorporation in products they ultimately buy back. The onus is on the issuer to check quality, but this does not absolve us from knowingly incorporating poor quality components into finished product.
PRODUCT IDENTIFICATION AND TRACEABILITY
Clause 4.8 This clause deals with product identification and traceability. A manufactured product must be traced through the various stages of its production and delivery. This is crucial to help identify the stage and hence the cause of any product failure. Where different products are similar or identical they must be identified using a method of labeling or coding. Note: In the case of a mail-order/telesales business this clause may apply to mail, checks, registration forms, etc. Clause 4.9 The company must be able to demonstrate that the overall process (sales right through to delivery to the end user) is in control.
There are many significantly different processes in both the manufacturing and service sectors. Accordingly only principles are set out in Clause 4.9, details having to be put in place by each organization. The main point is that process steps need to be identified and a plan to implement needs to be declared and documented.
The process must be carried out in a controlled way, so that plans / procedures / instructions are to hand, equipment and personnel used are adequate and that the general conditions are amenable to achieving the plans. To ensure that a process is carried out in a controlled manner the process must be well documented and the staff well trained. Many changes have been made in the 1994 version of ISO 9002 This clause addresses inspection and testing for incoming goods, in-process and final inspection.
Inspection of incoming materials must be directed by a sampling plan which details the selection of materials for inspection / testing from each received batch. Results must be documented, describing material inspected, number of rejects, reasons for rejection and corrective action. In-process inspection similarly needs to be dealt with, tying in with clause 4.8 “Product Identification and Traceability”.
Upon completion all products must undergo clearly specified final inspection. NOTE: Sub-clause 4.10.2.2 is a new addition and reads as follows… “In determining the amount and nature of receiving inspection, consideration shall be given to the amount of control exercised at the subcontractor’s premises and the recorded evidence of conformance provided”. This deals with the practice of ship-to-stock based on certificates of conformance or other recorded evidence of conformance of product or service.
Clause 4.11 This clause may not apply if test and measuring is not used, but make sure not to overlook the fact that items such a weighing scales require calibration and that in the past some auditors have required that steel rules be calibrated..
CONTROL OF INSPECTION, MEASURING AND TEST EQUIPMENT
This clause covers inspection, measuring and test equipment. It deals with the equipment used in inspection and testing, ensuring that it is suitable for its purpose. Frequency of calibration of equipment used in manufacturing should be based on stability, purpose and degree of utilization. Records of maintenance work must be retained. Recalibration must be in line with official standards or , where this is not the case, with a clearly documented method. 6 of 10 9/28/2009 1:28 PM In order to satisfy this part of the requirement, we must ensure that all equipment used to verify or measure product is subjected to our management control system. All this equipment must be identified and registered and we must have a process for putting all newly acquired equipment on the register.
WE MUST ENSURE THAT:
A calibration and maintenance system is in place. A starting point is a calibration register of equipment coupled with the location capital asset register. Trained personnel are available to operate the necessary procedures. No unregistered or overdue equipment is used. Evidence is provided which confirms that procedures are effective. All weighing machines are calibrated. Procedures allow for the use of personal equipment or equipment on loan or trial. Equipment which does not need to be calibrated is identified.
Clause 4.12 This clause deals with inspection and test status. There needs to be an indicator that states what tests a product has been through and whether it passed or failed. In order to satisfy this part of the requirement, the company must: Have a system or method for identifying the inspection and test status of the product at all relevant stages in the process. Ensure that the system used is defined as part of the process specification or is written documented work instructions.
Ensure that the personnel who operate processes related to this segregation system are trained in the discipline required to maintain it. Ensure that only products that have passed the required inspection and tests are shipped, put into use, or installed. This clause also refers to sub-clause 4.13.2 “
Review and Disposition of Non-conforming Products.” The responsibility for review and authority for the disposition of non-conforming product shall be defined. Non-conforming material shall be reviewed in accordance with documented procedures and it may be: Reworked to meet the specified requirements: Accepted with or without repair or concession: Regarded for alternative applications: Rejected or scrapped. Clause 4.13 7 of 10 9/28/2009 1:28 PM This clause stresses the control of non-conforming product. The aim here is to ensure that non-conforming product is not used by mistake.
Non-conforming material must be isolated and clearly identified. Every non-conforming lot should be accompanied by paperwork detailing the reasons for failure. Defects and claims should be sent to vendors without delay. Clause 4.14 This clause provides for detailed and timely documentation of corrective actions taken. Time must be taken to learn from mistakes but, strictly speaking, clause 4.14 wants avoidance of mistakes to be achieved following analysis of quality records, service reports and customer complaints. In all cases corrective action must be monitored. To satisfy ISO 9002 requirement 4.14, the Company must: Establish and document a system for investigating the cause of defective products or processes.
Establish a system for ensuring that appropriate corrective action is decided upon and implemented. Ensure that responsibility for corrective action is clearly defined. Establish and document procedures for analyzing the business. Keep records of all complaints and follow-up actions. Correct any deficiencies before they can cause defects in products or processes. Establish and document a process to ensure that corrective actions are implemented in an effective manner. Keep records of defects, the investigation of their cause and the corrective actions. Establish and document a process that ensures procedure amendments, resulting from corrective actions, are recorded and that work methods are changed to reflect the changes in amended procedures.
Clause 4.15 A policy must be set for handling, storage and packaging. Handling of products, i.e. when loading and unloading, must be done with due care for the product. Storage areas must be clean and the environment properly controlled (for example, heat, humidity, light.) Access to storage areas should be controlled. Controls should be implemented to identify slow moving, obsolete, deteriorated and damaged items (some of these may not apply in all cases but scrap should not be allowed to accumulate).
The company also needs to ensure the protection of the quality of product after final inspection and test. Clause 4.16 This clause covers quality records and is of an administrative nature. Quality records are the routine control and reporting documents that show the progress of the quality management system. Quality records should be kept over a reasonable length of time and stored in a safe secure area. The following are key records to be kept: Audits of the quality system. Calibration of test and measuring equipment Analyses of process control data. Records of corrective actions. Customer complaints.
Records of concessions (deviations). Training records. NOTE: Records may be in the form of any type of media, such as hard copy or electronic media. NOTE: Procedures must exist for backup / security of all records on electronic media. Clause 4.17 Planned and documented checks on the implementation and operation of the Quality Management System help to confirm that it is being operated correctly and effectively. Internal Quality Audits must be conducted on a regular basis. Coverage, schedules and standard procedures for each audit must be pre-determined. Records must be kept, such as the following: Deficiencies found. Corrective action required. Time agreed for corrective action to be carried out. Personnel responsible for corrective action.
NOTES: The auditor(s) must be independent of the areas being audited. The results of such audits must form an integral part of the input to management review activities. 9 of 10 9/28/2009 1:28 PM Clause 4.18 Clause 4.18 underlines the need for training. Training of personnel is concerned with the competence of personnel to do their assigned task. Where a lack of skills of qualifications is found, staff need to attend internal or external training courses. If a member of staff is new to the task then efforts must be made to train them. Staff performance needs to be regularly appraised and recorded.
Know-how must be kept up to date enabling staff to operate adequately the equipment that they are responsible for. Clause 4.19 Clause 4.20 Clause 4.20 stresses the value of measurement using statistical techniques. Statistical techniques should be used to assess the following: Reliability such as longevity and durability. Process control / capability studies. Determining quality levels and thus inspection plans.
Data analysis, performance assessment and defect analysis. 4.20.1 Identification of Need. The term “where appropriate” has been deleted from this clause and the supplier is required to identify the need for statistical techniques in order to control and verify process capability and product characteristics. 4.20.2 Procedures. The supplier shall establish and maintain documented procedures to implement and control the statistical techniques identified.