DNA is a natural polymer. It was first isolated in 1869 by Meischer, but its role in determining heredity remained unrecognized until 1944, by which time it was appreciated that it is the chromosomes within a cell nucleus that The word ‘theory’ comes from the Greek theoreo, meaning ‘I look at’. A theory is something we look at, pending acceptance or rejection. dictate hereditary traits. And such chromosomes consist of DNA and protein. In 1944, the American bacteriologist Oswald Avery showed how it was the DNA that carried genetic information, not the protein.
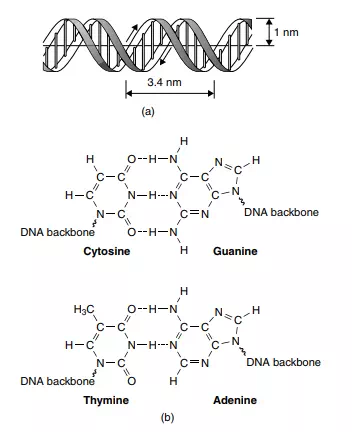
The next breakthrough came in 1952, when Francis Crick and Donald Watson applied X-ray diffraction techniques to DNA and elucidated its structure, as shown schematically in Figure 2.5. They showed how its now famous ‘double helix’ is held together via a series of unusually strong dipole–dipole interactions between precisely positioned organic bases situated along the DNA polymer’s backbone. There are four bases in DNA: guanine, thymine, cytosine and The rules of ‘base pairing’ (or nucleotide pairing) in DNA are: adenine (A) always pairs with thymine (T); cytosine (C) always pairs with guanine (G). adenine. Each has a ketone C=O group in which the oxygen is quite electronegative and bears an excess negative charge δ−, and an amine in which the electropositive hydrogen atoms bear an exces

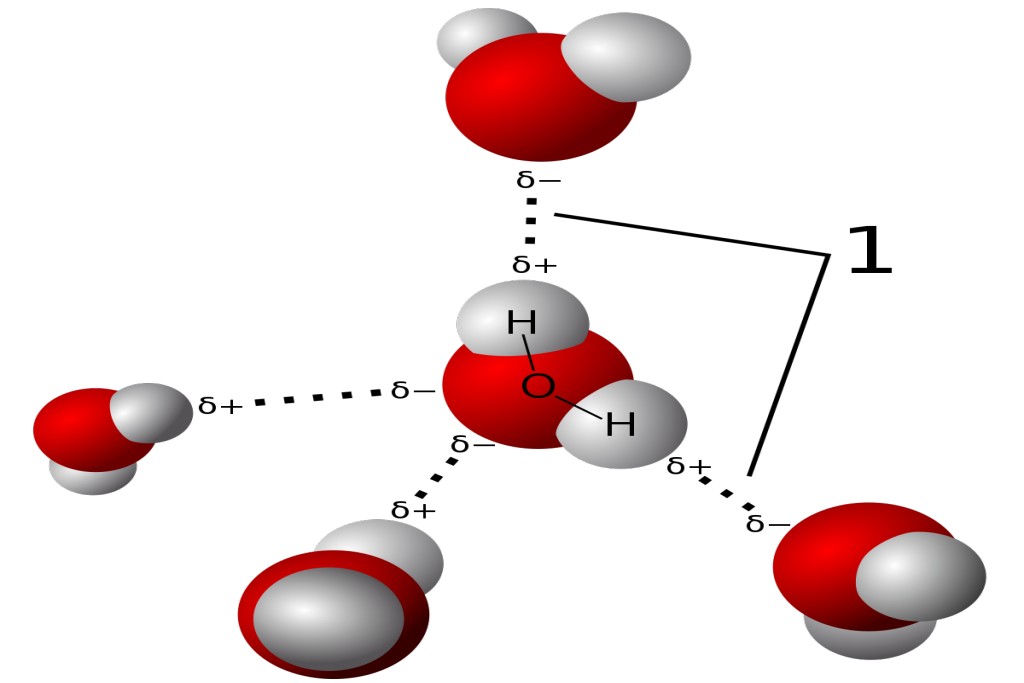
We call these extra-strong dipole–dipole bonds ‘hydrogen bonds’, IUPAC is the International Union of Pure and Applied Chemistry. It defines terms, quantities and concepts in chemistry. and these are defined by the IUPAC as ‘a form of association between an electronegative atom and a hydrogen atom attached to a second, relatively electronegative atom’. All hydrogen bonds involve two dipoles: one always comprises a bond ending with hydrogen; the other terminates with an unusually electronegative atom.
It is best considered as an electrostatic interaction, heightened by the small size of hydrogen, which permits proximity of the interacting dipoles or charges. Table 2.2 contains typical energies for a few hydrogen bonds. Both electronegative atoms are usually (but not necessarily) from the first row of the periodic table, i.e. N, O or F. Hydrogen bonds may be intermolecular or intramolecular. Finally, as a simple illustration of how weak these forces are, Strictly, the dipole– dipole interactions discussed on p. 42 are also hydrogen bonds, since we discussed the interactions arising between H–O-bonded species. note how the energy required to break the hydrogen bonds in liquid hydrogen chloride (i.e. the energy required to vaporize it) is 16 kJ mol−1 , yet the energy needed break the chemical bond between atoms of hydrogen and chlorine in H–Cl is almost 30 times stronger, at 431 kJ mol−1 .