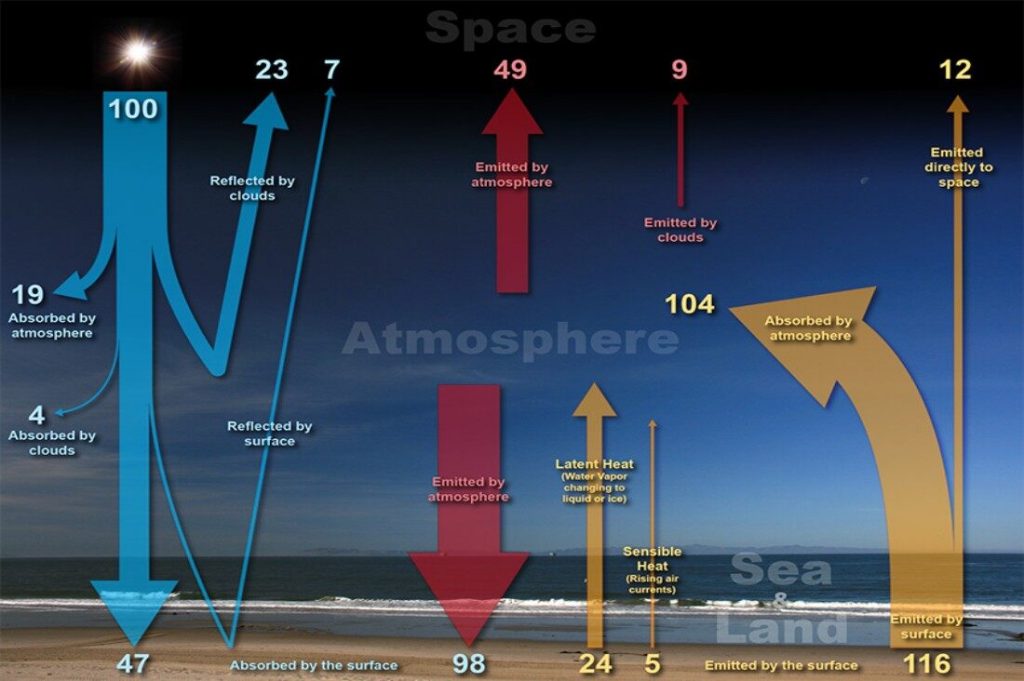
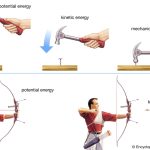
The concept of energy conservation as expressed by an energy balance equation is central to chemical engineering calculations. Similar to mass balances studied previously, a balance on energy is crucial to solving many problems.
System
A “system” is an object or a collection of objects that an analysis is done on. The system has a definite boundary, called the system boundary, that is chosen and specified at the BEGINNING of the analysis. Once a system is defined, through the choice of a system boundary, everything external to it is called the surroundings. All energy and material that are transferred out of the system enter the surroundings, and vice versa.
In the general case there are very few restrictions on what a system is; a system can have a nonzero velocity, a nonzero acceleration, and a system can even change in size with time. An isolated system is a system that does not exchange heat, work, or material with the surroundings. If heat and work are exchanged across a system’s boundary, but material is not, it is a closed system. An open system can exchange heat, work, and material with the surroundings.
Examples. Discuss each situation below as approximating an isolated, a closed, or an open system.
(i) A river.
(ii) The interior of a closed can of soda.
(iii) The interior of a closed refrigerator that is turned on.
(iv) The interior of a closed refrigerator that is turned off.
State of a System
Once a system is defined, a certain number of variables will specify its state fully. For example, one may need to provide the temperature, pressure, composition, total amount of material, velocity, and position in order to specify a system’s “state.” The exact information that is needed to specify the state of a system depends on the type of system and the analysis to be performed.
State Functions and State Properties
The state of a system can be changed, for example by increasing its temperature or changing its composition. Properties of the system whose change depends only on the initial (before) and final states of the system, but not on the manner used to realize the change from the initial to the final state, are referred to as state properties or state functions. In other words, the change in a state function or state property X, between some final (state 2) and initial (state 1) situations, can be expressed as
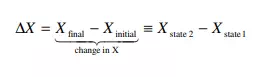
In equation 1, Xfinal only depends on the final state of the system, and Xinitial only on the initial state of the system. Equation 1 does not require any information whatsoever as to how the system got from the initial to the final state, since X does not depend on the details of the path followed. Example. Which of the below examples represent changes in state functions?
(i) Work done to climb from the bottom (state 1) to the top (state 2) of a mountain.
(ii) Change in gravitational energy of an object when it is raised from the bottom (state 1) to the top (state 2) of a mountain.
(iii) Change in density of water in a pot when it is heated from 20 oC (state 1) to 50 oC (state 2).
(iv) Amount of heat liberated from burning gas in a stove in order to realize a temperature change of the water in a pot from 20 oC (state 1) to 50 oC (state 2). (This requires some thought …)